Fluorescein Sodium Ophthalmic Strips – SFDA Approved
- Estimated Delivery : ±2 business days Inside Saudi Arabia
In modern ophthalmology, accuracy and efficiency are essential for proper diagnosis and patient care. Fluorescein Sodium Ophthalmic Strips play a critical role in detecting corneal abrasions, evaluating tear film stability, and supporting a wide range of eye examinations. With increasing demand in Saudi Arabia, healthcare providers are seeking products that combine quality, regulatory compliance, and consistent availability.
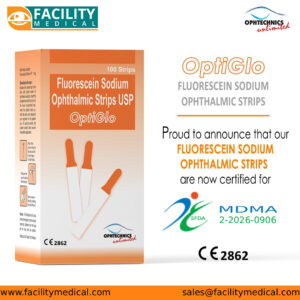
OptiGlo Fluorescein Sodium Ophthalmic Strips, supplied by Facility Medical, are now officially SFDA registered (MDMA: 2-2026-0906), making them a trusted and compliant solution for hospitals, clinics, and eye care professionals across the Kingdom.
These ophthalmic strips are designed to deliver precise and uniform fluorescein staining, ensuring clear visualization during diagnostic procedures. Each strip is manufactured under strict quality standards to guarantee sterility, safety, and consistent performance. This is particularly important in busy clinical environments where reliability directly impacts patient outcomes.
One of the key advantages of OptiGlo is its dual certification. In addition to SFDA approval, the product is also CE certified (CE 2862), reflecting adherence to international medical device standards. This combination gives healthcare providers confidence in both local regulatory compliance and global quality benchmarks.
Supply chain reliability is another major concern in the medical sector. Facility Medical addresses this by ensuring stable stock availability and fast delivery, helping clients avoid delays that could disrupt operations. This makes OptiGlo a strong and dependable alternative to other competitors in the Saudi market, particularly when consistency and responsiveness are required.
From routine eye exams to specialized ophthalmic procedures, OptiGlo Fluorescein Strips are suitable for:
-
Corneal injury detection
-
Tear film break-up time (TBUT) testing
-
Contact lens fitting assessments
-
General ophthalmic diagnostics
By choosing a locally registered and internationally certified product, healthcare providers can streamline procurement processes, reduce regulatory risks, and maintain high standards of patient care.
Facility Medical remains committed to delivering high-quality ophthalmic solutions tailored to the needs of the Saudi healthcare market. With OptiGlo, you gain a product that combines precision, compliance, and availability—all in one solution.
| GENERAL INFORMATION | |
|---|---|
| Authorized Representative | Facility Medical Company || ARL-2026-MD-0056 |
| MDMA | MDMA: 2-2026-0906 |
| Product Name | OptiGlo || Fluorescein Sodium Ophthalmic Strips |
| Manufacturer | Ophtechnics Unlimited |
| Indications For Use | Ophthalmic diagnostic procedures |
| Active Ingredient | Fluorescein Sodium |
| Pack Size | 100 Strips per Box |
| Supplier | Facility Medical Company |
| Shelf Life | 5 Years |
| Format | Sterile Ophthalmic Strips |

















Reviews
There are no reviews yet.